|
In this equation, A is the current in amperes and C the charge in coulombs. A concentration cell acts to dilute the more concentrated solution and concentrate the more dilute solution, creating a voltage as the cell reaches an equilibrium. Notice that both sides are both charge balanced and atom balanced.\nonumber \] A concentration cell is an electrolytic cell that is comprised of two half-cells with the same electrodes, but differing in concentrations. H +, H 2O, and e - can be used to balance the charges and atoms in acidic conditions. OH -, H 2O, and e - can be used to balance the charges and atoms in basic conditions. This is done by adding H 2O, OH -, e -, and or H + to either side of the reaction until both atoms and charges are balanced. Due to this electrolyte it may be more difficult to satisfy the balance of both the atoms and charges. It is also possible and sometimes necessary to consider a half-reaction in either basic or acidic conditions, as there may be an acidic or basic electrolyte in the redox reaction. and finally becomes Cl 2 + 2Fe 2 + → 2Cl - + 2Fe 3 +.A chemist can atom balance and charge balance one piece of an equation at a time. 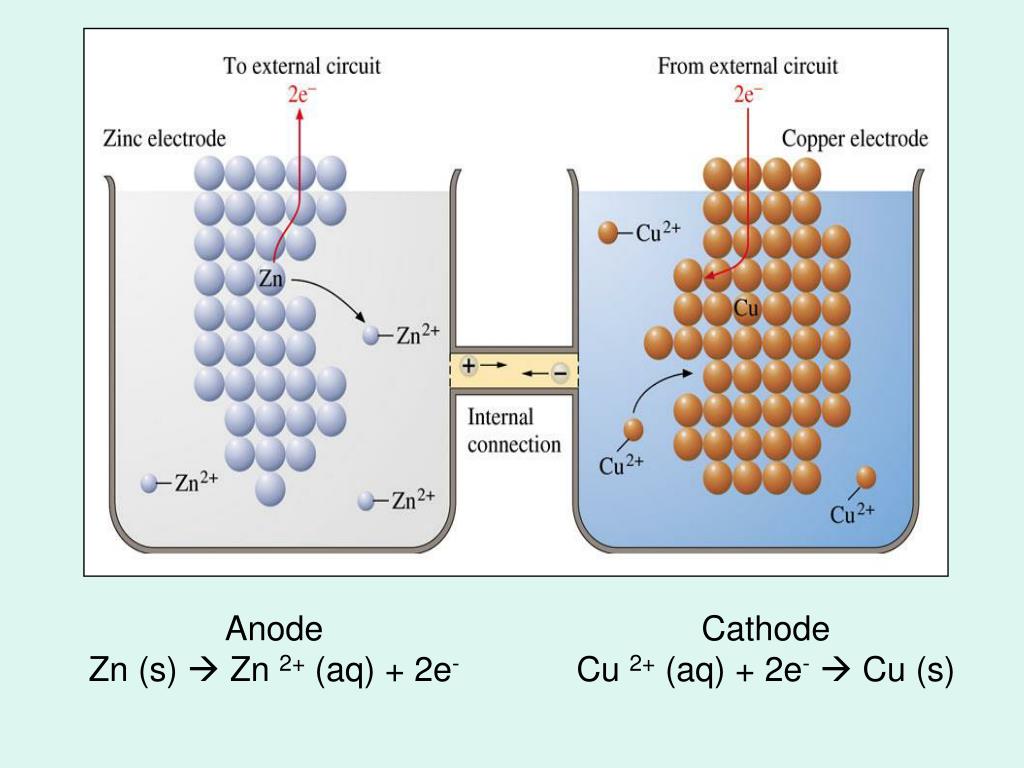
Decomposition is also a way to simplify the balancing of a chemical equation. Note the transfer of electrons from Fe to Cl. For example, in the above reaction, it can be shown that this is a redox reaction in which Fe is oxidised, and Cl is reduced. The decomposition of a reaction into half-reactions is key to understanding a variety of chemical processes. Given two half-reactions it is possible, with knowledge of appropriate electrode potentials, to arrive at the full (original) reaction the same way. These changes can be represented in formulas by inserting appropriate electrons into each half-reaction: Fe 2 + → Fe 3 + + e − Cl 2 + 2e − → 2Cl − There are then effectively two half-reactions occurring. The two elements involved, iron and chlorine, each change oxidation state iron from +2 to +3, chlorine from 0 to -1. The sum of these two half-reactions is the oxidation- reduction reaction.Ĭonsider the reaction below: Cl 2 + 2Fe 2 + → 2Cl − + 2Fe 3 + In any given oxidation-reduction reaction, there are two half-reactions – oxidation half- reaction and reduction half-reaction. Two ions, positive (Mg 2 +) and negative (O 2-) exist on product side and they combine immediately to form a compound magnesium oxide (MgO) due to their opposite charges (electrostatic attraction). After canceling, the equation is re-written as 2Mg(s) + O 2(g) →2Mg 2 + + 2O 2. Due to this, electrons appearing on both sides of the equation are canceled. What we see is the reactants (starting material) and end products. When chemical reaction, especially, redox reaction takes place, we do not see the electrons as they appear and disappear during the course of the reaction. A galvanic cell (left) transforms the energy released by a spontaneous redox reaction into electrical energy that can be used to perform work. The overall reaction is the sum of both half-reactions: 2Mg(s) + O 2(g) + 4e − →2Mg 2 + + 2O 2 - + 4e − Thus, a reduction half-reaction can be written for the O 2 as it gains 4 electrons: O 2(g) + 4e − → 2O 2. On the other hand, O 2 was reduced: its oxidation state goes from 0 to -2. Since there are 2 Mg on left side, a total of 4 electrons are lost according to the following oxidation half reaction: 2Mg(s) → 2Mg 2 + + 4e − 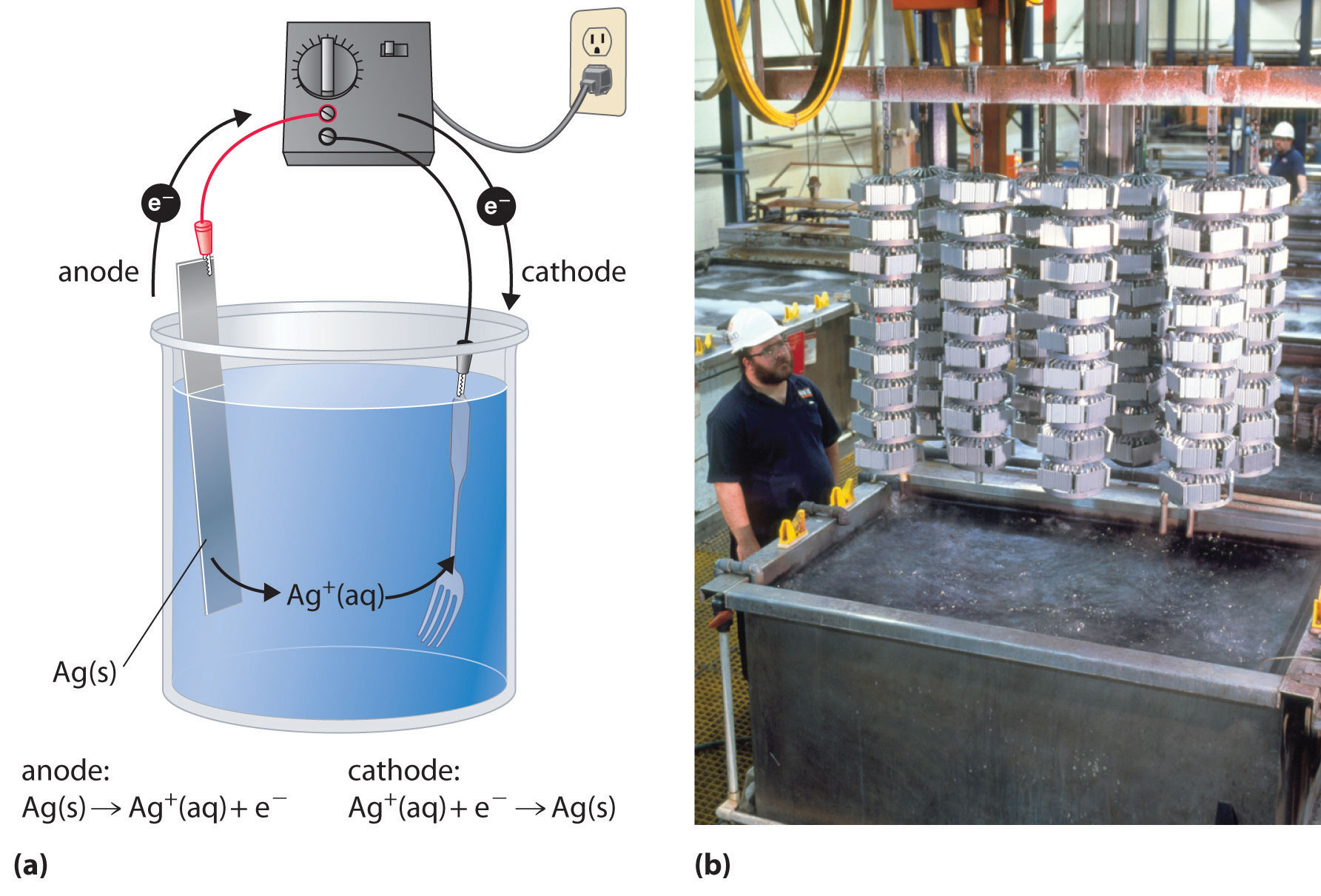
This is because when Mg(s) becomes Mg 2 +, it loses 2 electrons. The Mg(s) with zero charge gains a +2 charge going from the reactant side to product side, and the O 2(g) with zero charge gains a -2 charge. Magnesium oxide is an ionic compound containing Mg 2 + and O 2- ions whereas Mg(s) and O 2(g) are elements with no charges. When magnesium burns, it combines with oxygen (O2) from the air to form magnesium oxide (MgO) according to the following equation: 2Mg(s) + O 2(g) → 2MgO(s) Photograph of a burning magnesium ribbon with very short exposure to obtain oxidation detail.Ĭonsider the example burning of magnesium ribbon (Mg). For oxidation-reduction reactions in basic conditions, after balancing the atoms and oxidation numbers, first treat it as an acidic solution and then add OH - ions to balance the H + ions in the half reactions (which would give H 2O). For oxidation-reduction reactions in acidic conditions, after balancing the atoms and oxidation numbers, one will need to add H + ions to balance the hydrogen ions in the half reaction. Half-reactions are often used as a method of balancing redox reactions. Half-reactions can be written to describe both the metal undergoing oxidation (known as the anode) and the metal undergoing reduction (known as the cathode). 
Often, the concept of half-reactions is used to describe what occurs in an electrochemical cell, such as a Galvanic cell battery. A half reaction is obtained by considering the change in oxidation states of individual substances involved in the redox reaction. \)Ī half reaction is either the oxidation or reduction reaction component of a redox reaction.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
